HOME|Our Favorites
HEC Pharm
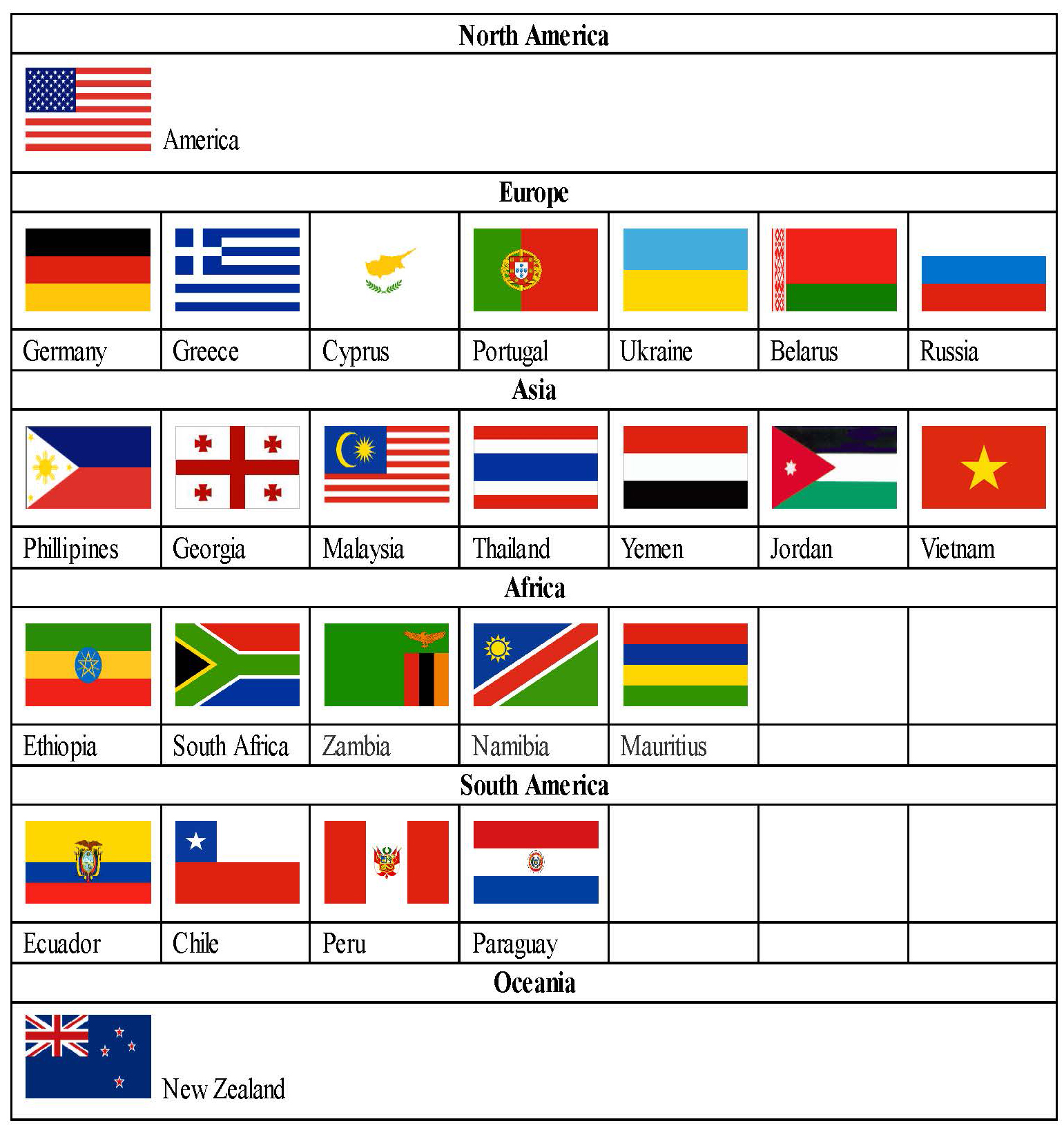
Product Name | Standard | Strength | Packing Size | Status |
Amlodipine besylate Film-coated tablet | USP | 2.5/5/10mg | 90s x 1 | Global marketing |
In-house | 5/10mg | 10s x 1 | ||
Azithromycin 1H2O Film-coated tablet | USP | 250/500/600mg | 30s x 1 | US under review |
Azithromycin 2H2O Film-coated tablet | In-house | 250mg 500mg | 6s x 1 3s x 1 | Global marketing |
Ciprofloxacin Film-coated tablet | In-house | 100/250/500/750mg | 10s x 1 | Global marketing |
Clarithromycin Film-coated tablet | USP | 250mg/500mg | 60s x 1 | |
In-house | 250mg 500mg | 5s x3 7sx2/6, 10sx2 | ||
Clarithromycin Extended-release tablet | USP | 500mg | 10s x 1 | Global marketing |
In-house | 500mg | 10s x 1/2/10 | ||
Entacapone Film-coated tablet | USP | 200mg | 60s x 1 | Global marketing |
In-house | 200mg | 30s x 1 | ||
USP In-house | 20/40mg | 60s x 1 | EU approving in 2017 | |
Fingolimod Capsules | In-house | 0.5mg | 7s x 1/4 | US Under review |
Ibuprofen Film-coated tablet | USP | 400/600/800mg | 100/60/50s x 1 | US Under review |
Irbesartan Film-coated tablet | USP | 75/150 300mg | 30/90s x 1 30s x 1 | Global marketing |
In-house | 75/150/300mg | 6s x 1 | ||
Levofloxacin Film-coated tablet | 250/500/750mg | 50/50/20s x 1 | Global marketing | |
In-house | 250/500mg | 6/10s x 1 | ||
Linagliptin Tablets | In-house | 5mg | 30s x 1 | US Under review |
Linagliptin and Metformin Hydrochloride Tablets | In-house | 2.5mg/500mg 2.5mg/850mg 2.5mg/1000mg | 60s x 1 | US Under review |
Metformin tablets | USP | 500/850/1000 | 1000s x 1 | US Under review |
Moxifloxacin HCl Film-coated tablet | USP | 400mg | Global marketing | |
In-house | 400mg | 10s x 1 | ||
Sitagliptin and Metformin Film-coated tablet | In-house | 50/850mg; 50/1000mg | 14/60 x 1 | EU approving in 2017 |
Olanzapine Film-coated tablet | 2.5/5/7.5/10/15/20mg | 30s x 1 | Global marketing | |
In-house | 2.5/5/7.5/10/15/20mg | |||
Olanzapine Orally Disintegrating Tablets | USP | 5/10/15/20mg | 30s x 1 | Global marketing |
In-house | 5/10/15/20mg | 10s x 1 | ||
Olmesartan medoxomil tablets | In-house | 10 /20 /40 mg | 7/10s x 1 | EU approving in 2017 |
Prasugrel HCl Film-coated tablet | 5/10mg | US Under review | ||
Rivaroxaban Tablets | 10/15/20mg | 30s x 1, 90s x 1 | US Under review | |
Film-coated tablet | USP | 5/10/20/40/80mg | 30s x 1 | US Under review |
Ticagrelor Tablets | In-house | 90mg | 60s x 1 | US Under review |
Zidovudine Film-coated tablet | USP | 300mg | Global marketing |
Product Name | Dosage Form | Standard | Strength | Packing Size | Status |
Azithromycin | Capsules | CP | 250 mg | 6s × 1/2 | Marketed |
Azithromycin | Dispersible Tablets | CP | 250 mg | 6s × 1/2 | Marketed |
Azithromycin | Suspension | CP | 100 mg | 6s, 8s, 12s | Marketed |
Amlodipine besylate | Tablets | CP | 5 mg | 7s, 14s, 28s | Marketed |
Benzbromarone | Tablets | CP | 50 mg | 10s | Marketed |
Cetirizine Hydrochloride | Dispersible Tablets | CP | 10 mg | 6s × 1/2/4 | Marketed |
Clarithromycin | Dispersible Tablets | CP | 250 mg | 6s | Marketed |
Clarithromycin | Tablets | CP | 250 mg | 6s | Marketed |
Fluconazole | Capsules | CP | 50 mg | 6s, 10s | Marketed |
Fudosteine | Tablets | CP | 200mg | 6s× 2 | Marketed |
Glipizide | Capsules | CP | 5mg | 10s × 3 | Marketed |
Levofloxacin lactate | Tablets | CP | 100 mg | 10s | Marketed |
Lisinopril | Tablets | CP | 10 mg | 7s × 1/2/4 | Marketed |
Mycophenolate mofetil | Powder for injection | CP | 500mg | 2s | Marketed |
Oxaprozin | Enteric-coated tablets | CP | 200 mg | 8s × 1/2/3 | Marketed |
Ozagrel sodium | Powder for injection | CP | 20mg | 8s | Marketed |
Simvastatin | Tablets | CP | 10 mg | 10s | Marketed |
20 mg | 7s × 1/2 | Marketed | |||
Telmisartan | Tablets | CP | 40 mg,80 mg | 7s × 1/2 | Marketed |
Valacyclovir Hydrochloride | Tablets | CP | 300 mg | 6s, 10s | Marketed |
Copyright HEC Pharma Co., Ltd license key:粤ICP备19111996号